Recently, the research team led by Professor Youjun Zhang from our institute published a research article titled "The pleiotropic effects of a single transcription factor underpin trade-offs associated with insecticide resistance in whitefly" in the renowned journal Journal of Advanced Research (IF=12.99). This study elucidates the molecular mechanism by which the GPCR-MAPK-E75 signaling axis regulates the trade-off between neonicotinoid insecticide (thiamethoxam) resistance and reproduction in Bemisia tabaci, providing a theoretical basis for resistance monitoring, early warning, and integrated management of this vegetable pest.

Bemisia tabaci (whitefly) is a major vegetable pest that poses a serious threat to crop production in China and worldwide. Neonicotinoid insecticides are the primary agents currently used to control whitefly in the field; however, long-term excessive application has led to high levels of resistance. Previous research by the team identified that enhanced detoxification metabolism mediated by cytochrome P450s is the dominant mechanism underlying insecticide resistance in field populations of whitefly in China. Nevertheless, resistance evolution is often accompanied by fitness costs such as developmental delay and reduced fecundity. This "benefit-cost" trade-off is not only a critical factor in resistance evolution but also serves as an important basis for formulating resistance management strategies.
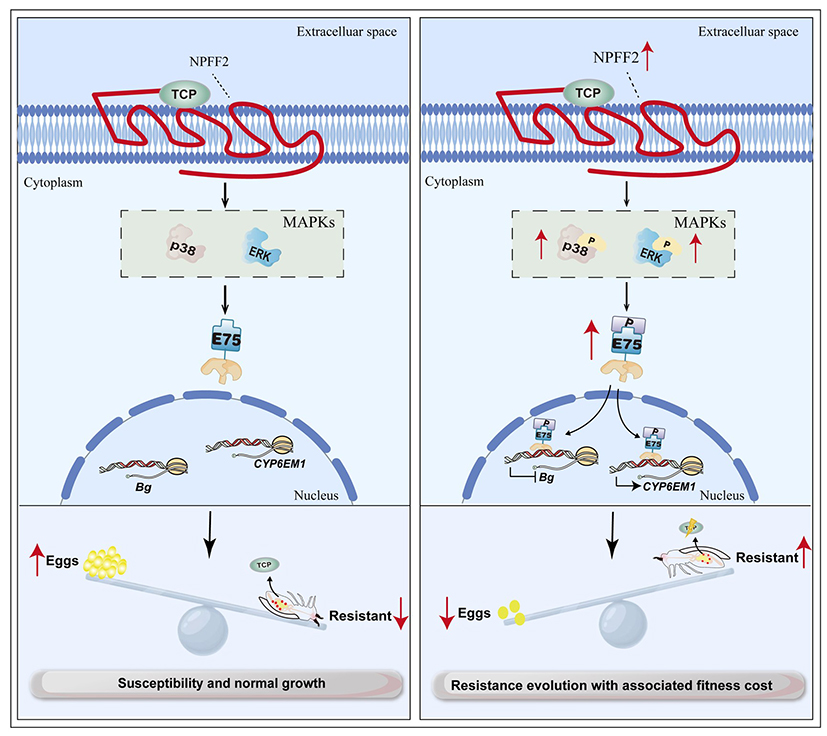
In this study, using insecticide bioassays and age-stage, two-sex life tables, the team determined the resistance level of whitefly to the neonicotinoid insecticide thiamethoxam and the associated fitness cost of reduced fecundity. Furthermore, through Western blot, immunofluorescence, molecular docking and dynamics simulations, recombinant P450 expression, and UPLC-MS/MS analysis, the function of the cytochrome P450 enzyme CYP6EM1 in thiamethoxam resistance was clarified. Additionally, using dual-luciferase reporter assays, Phos-tag phosphorylation analysis, EMSA, yeast one-hybrid, and co-immunoprecipitation assays, the study revealed that the phosphorylated and activated ecdysone-inducible protein 75 (E75) can simultaneously bind to the promoter regions of both the resistance-associated P450 gene CYP6EM1 and the gene Bg implicated in the reproductive cost, leading to increased resistance levels accompanied by reduced fecundity as a fitness cost. The study also identified that the GPCR protein NPFF2 activates the MAPK (p38 and ERK) signaling pathway, promoting the phosphorylation of downstream E75. This elucidates that the GPCR-MAPK-E75 regulatory axis exerts dual transcriptional regulatory functions: positively activating the expression of the detoxification P450 gene CYP6EM1, while negatively suppressing the transcription of the key oogenesis gene Bg.
This paper elucidates the molecular mechanism by which the GPCR-MAPK-E75 signaling axis regulates the trade-off between thiamethoxam resistance and reproductive fitness in whitefly. It confirms that E75 acts as a "molecular switch," achieving dual regulation—activation of resistance and suppression of reproduction—by differentially binding to target gene promoters. It also validates that the GPCR-MAPK pathway serves as a conserved upstream hub regulating the fitness trade-off of insecticide resistance. This study enriches our understanding of the transcriptional regulatory network underlying insecticide resistance and provides a theoretical foundation for developing novel pest management strategies targeting detoxification enzyme signaling pathways.
Postdoctoral researchers Mingjiao Huang, Shaonan Liu, Buli Fu, and Peipan Gong from our institute are the co-first authors of this paper. Professor Youjun Zhang and Researcher Xin Yang are the corresponding authors. This research was supported by grants from the National Natural Science Foundation of China and the Agricultural Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences.
Original link: https://doi.org/10.1016/j.jare.2026.03.033